Drop. Dissolve. Apply.
Three steps. No measuring. No mixing. No margin for error.


01
DROP
One tablet. One liter. 1,000 ppm. Every time.
Drop one CHLOR-CLEAN tablet into one liter of water. The effervescent tablet dissolves and delivers exactly 1,000 ppm available chlorine. No measuring, no mixing, no guesswork.2

02
DISSOLVE
Chlorine and detergent in a single solution.
The effervescent tablet dissolves completely into water, releasing NaDCC chlorine and integrated detergent. Combined cleaning and disinfection in one step. No concentrates, no measuring.

03
APPLY
Clean and disinfect. No rinse.
Wipe or mop. The integrated detergent lifts soil while the chlorine deactivates pathogens. 10 to 15 minutes contact time depending on the target pathogen and concentration.3
The process is the same at 1,000 ppm and at 5,000 ppm. The only thing that changes is the number of tablets.
One product. Every concentration. Every surface. Every shift.
The pathogens that matter. The standards that prove it.
Every satisfactory audit started the same way. Someone specified the right product at the right concentration. CHLOR-CLEAN is tested against the full pathogen spectrum using the European standards your protocol demands.
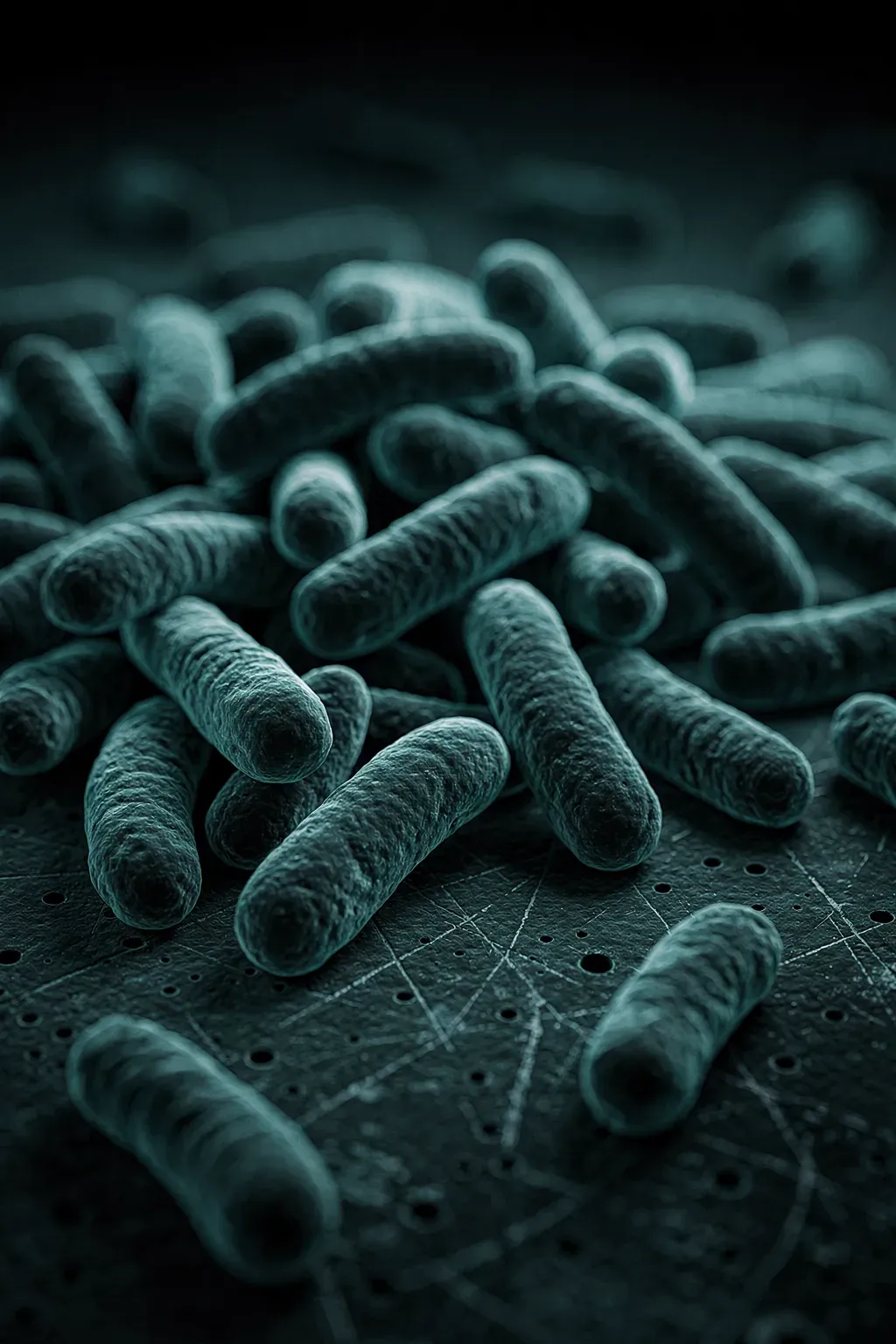
Bacteria
Proven across food, industrial, institutional and medical test scopes. The standard for ward cleaning, high-touch surfaces, kitchens, bathrooms, and general environmental disinfection.
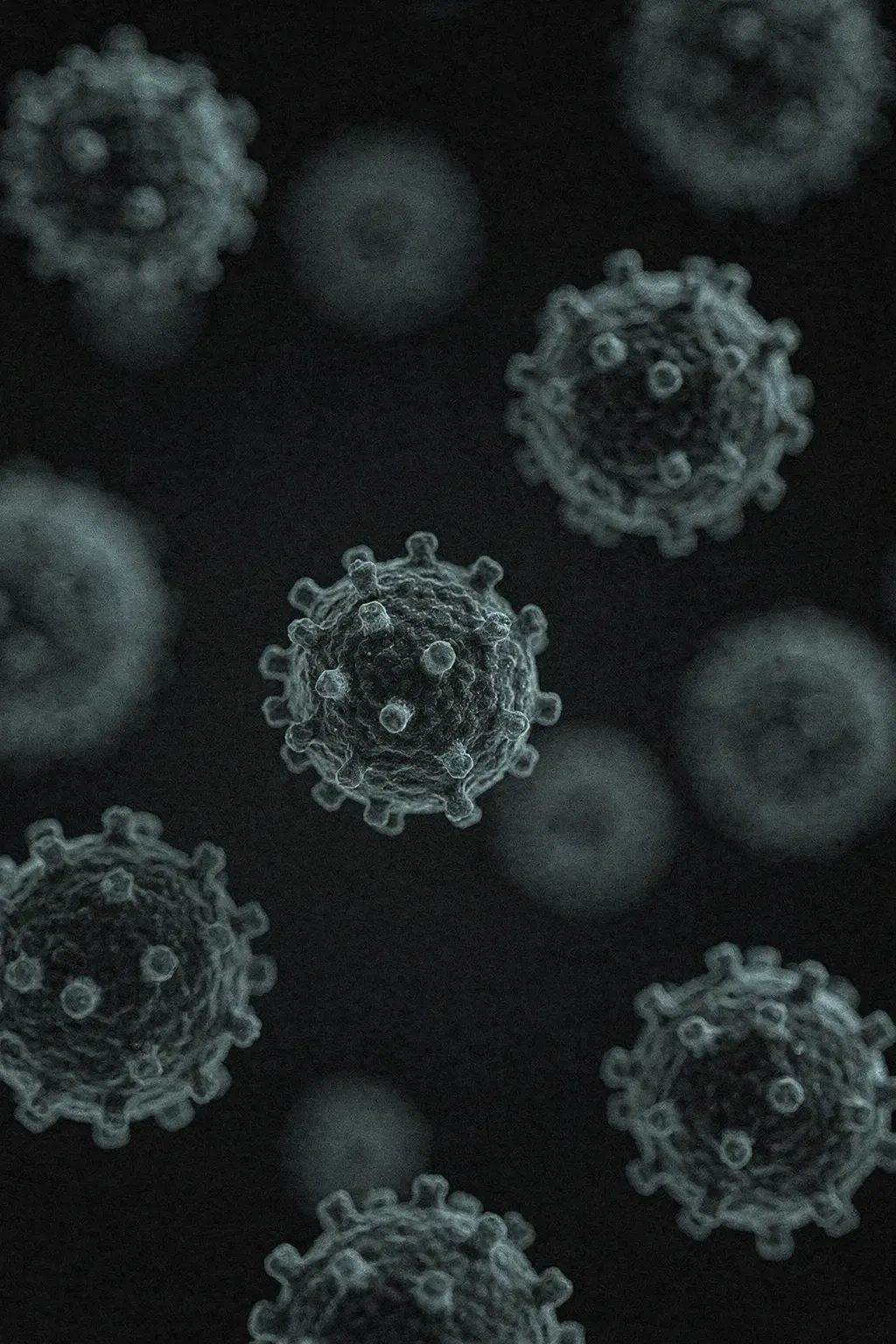
Viruses
Proven virucidal against enveloped and non-enveloped viruses. The product your norovirus outbreak protocol, respiratory virus response, and virucidal compliance requirement calls for.
Fungi
Proven fungicidal and yeasticidal to medical-area standards. Built for shower rooms, changing facilities, damp environments, and food-preparation areas where mold is a contamination risk.
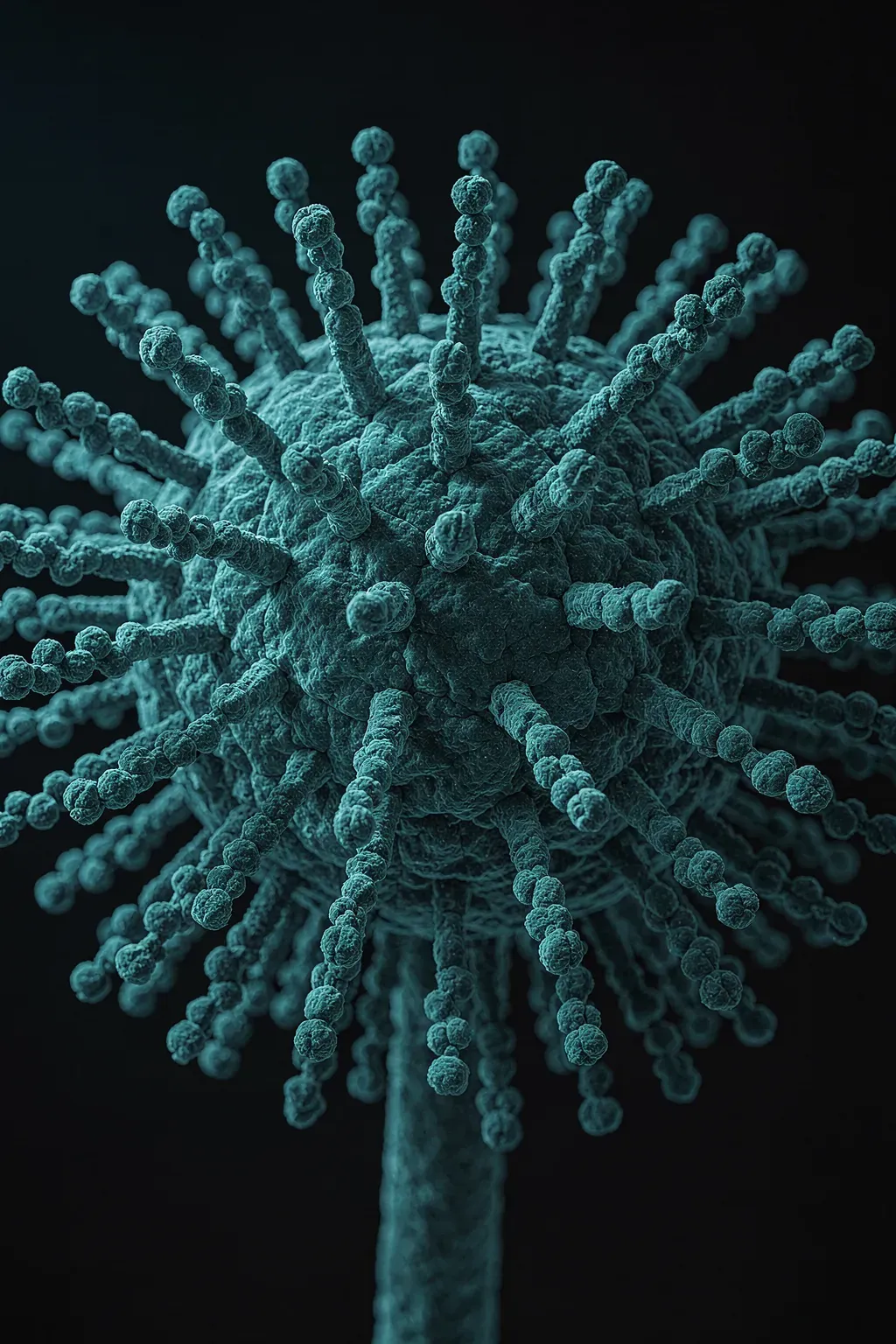
Spores
Proven sporicidal including against Clostridium difficile. The choice for outbreak response, isolation rooms, terminal cleans, and any facility where a sporicidal protocol is written into the SOP.

Mycobacteria
Proven mycobactericidal to the medical standard. Specified for tuberculosis isolation rooms and specialist clinical environments where mycobacterial contamination is a defined protocol risk.
You already know which pathogen keeps your IPC lead awake. We test against it.
Thirteen European standards. One tablet. The test data is in the documentation pack.
What your procurement team needs to know.
One product. One supplier. One training protocol.

One system
For your entire disinfection protocol
Switch without disruption
Your team gets trained before the first tablet is opened. Protocols are mapped to your existing workflows. There is no gap in coverage and no parallel procurement process. The transition runs on a defined schedule, and the team that formulated the product is the team that trains your staff.

50%+4
Lower total cost of disinfection
The cost drops because the steps disappear.
Your team currently cleans with a detergent, then disinfects with a separate product. CHLOR-CLEAN does both in one pass. That eliminates an entire labour round per surface, per room, per shift. One product also means one supplier, one SDS pack, one purchase order, and no separate fees for training or documentation.

Two weeks5
From first inquiry to operational deployment
Your first 90 days are mapped before you start
Your named account lead runs the on-site briefing in week one, checks compliance with your team in month one, and reviews the protocol with you in month three. One person owns the transition from your first call to business as usual.
Illustrative. Based on typical partnership outcomes. Individual results vary.
You are probably thinking your current contract is not up yet. That is fine. The conversation starts now. The switch happens on your timeline.
We build the proposal so your IPC lead, your finance director, and your board see the same numbers you just did. Onboarding partnerships for Q3 2026 are being scheduled now.
You are not buying a tablet. You are buying a system.
The tablet dissolves in two minutes. The system prevents infection for years.


Complete
The Documentation
Audit-ready. Current. Included.
SDS, occupational safety assessment, dilution protocol, contact time evidence. You hand the inspector one pack. Current. Complete.7

Direct
The Line
A scientist, not a call centre.
A spill protocol question. A surface you have never cleaned before. You call one number. Our in-house team picks up. No ticket. No hold music. No escalation. Direct.
The team that formulated the product is the team that trains your staff and owns your account.
There is no handoff. There is no call centre. There is a person with your name on their schedule.
Wherever hygiene is non-negotiable.
From hospital wards to school kitchens. One system, tailored to how your team actually works.

Hospitals and clinics
Ward cleaning, outbreak response, terminal disinfection. Your infection prevention team needs a product that works the same way every time, across every operator, every shift.

Long-term care and residential
Your cleaning team changes. Agency staff rotate. Your disinfection protocol cannot depend on who is on shift. One tablet in one liter of water. The same result every time.

Facilities
Schools, laboratories, public buildings. The compliance requirement is the same whether you manage one site or fifty. Same product, same protocol, same documentation pack.

Kitchens and food preparation
Commercial kitchens, canteens, food production. Where cross-contamination is not an option and your cleaning protocol must pass environmental health inspection every time.

Agriculture and veterinary
Livestock housing, milking parlours, hatcheries, veterinary surgeries, equine facilities. Where biosecurity is measured in pathogen kill rates, not marketing claims.
Your site has specific requirements. The protocol adapts. The product does not change.
Tell us about your environment and your location. We will tell you exactly how the system works there.
Trusted across 60+ countries.






















The next time someone questions your protocol, do you want a product label or a direct line to the team that formulated it?
Get pricing tailored to your organisation and a dilution guide built for your protocols. Speak directly with our in-house microbiologists and IPC leads either on-site at your facility or remotely.6 We'll cover dilution protocols, contact times, surface compatibility, and occupational safety documentation, all on your team's schedule.
Step 1 of 2
Thank you.
We have received your inquiry and will respond promptly with pricing tailored to your organisation.
